The rich, often “electric” rainbow colors of fluorite specimens from world-wide locations, the frequent appearance of different colors within regions, bound with the lattice planes within a crystal, and the sculptural appearances of stepped cubic and octahedral crystals, as shown in Figures 1-5, make fluorite a favorite among collectors.
In this blog I’ll describe those structural properties of fluorite that are relevant to lapidary practices, and the sources of its’ color, (Figure 8). I’ll describe how the regions, or zones, of different colors within a fluorite crystal develop. Also, because of their beauty, I am including a gallery of the beautiful fluorite specimens from around the world, that I found as I searched the web.





Properties of Fluorite [Ref 6]
Crystal Structure and Crystallography
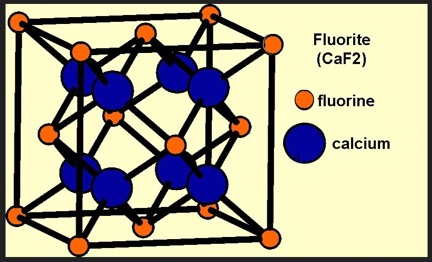
The crystal lattice of fluorite exhibits cubic symmetry with the calcium and fluorine ions positioned within the basic unit cell, as shown in Figure 6. The four fluorine ions F-1are positioned centrally in the unit cell with the calcium ions Ca2+distributed at the corners and the center of each face of the unit cell. In this array, each calcium ion has two fluorine ions as neighbors. Imperfections in the lattice underlie the presence of color in fluorite, as described below.
The lattice structure of fluorite results in its crystallizing in the isometric crystal system. Fluorite typically exhibits the basic crystal forms of the cube, octahedron, and rhombic dodecahedron shown in Figures 7 & 8. Fluorite also exhibits ball-like or grape-like botryoidal forms, but only rarely. (Figure 9)
The Mohs hardness of Fluorite is 4 and it exhibits a perfect cleavage on octahedral planes and less-well developed cleavage or parting on dodecahedral planes. A cleavage octahedron is shown in Figure 12 [Ref 14]. The ready cleavage and softness of fluorite requires the lapidarist to minimize the generation of stresses during the cutting and polishing of a stone.
Typical Crystal forms of Fluorite
Typical crystal forms are the cube, oxctahedron, and dodecahedron; fluorite also occurs in botryoidal form.


Mont Blanc, Chamonix, France [Ref 8]


Twinning in Fluorite
Fluorite forms both penetration twins [Ref 11] and Spinel Twins [Ref 12] on octahedral planes, as shown in Figures 10 and 11.


Cleavage in Fluorite

Colors in Fluorite
As shown in Figure 6, fluorite can occur in colorless form. Color, as described in Table I, is induced by the presence of an impurity ion, by nanoparticles of calcium metal, which scatter light preferentially according to size, or by inclusions of organic material such as bitumen.
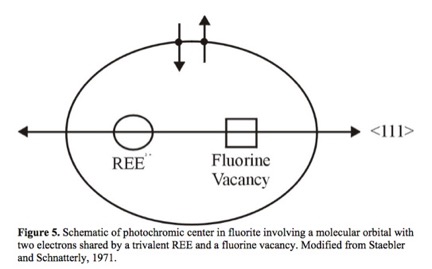
Ref 22. Page 27, Schematic of photochromic center color center–:
Color Zoning in Fluorite Crystals
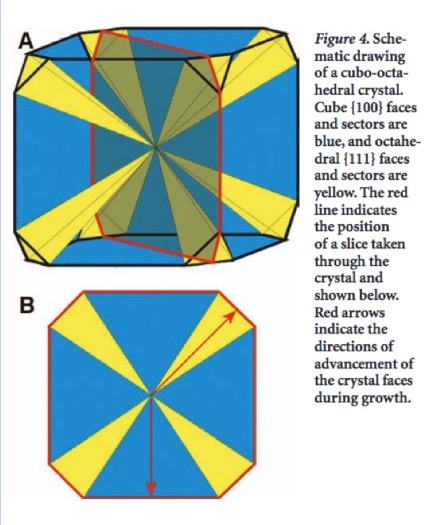
The establishment of discrete geometric zones of color in a fluorite crystal comes from the sequestration of a specific impurity, advancing along a crystallizing plane during sectorial growth [Ref 23]. A sketched example of sectorial growth in a fluorite crystal is shown in Figure 15, which is taken from Figure 4 of [Ref 24]. In the example, growth of octahedral and cubic planes are shown. In Figure11, showing a fluorite specimen from Namibia, growth is seen to have proceeded along both dodecahedral and cubic planes. Growth proceeded along advancing dodecahedral planes, forming the green diamond-shaped region, and along cubic planes, forming the deep green corner regions.

GALLERY OF FLUORITE SPECIMENS
In this Gallery I’ve included specimens from France, Morocco, Germany, Spain, and Mexico, which are not as well known to collectors as specimens from England the United States.











Ref 25. http://www.trinityminerals.com/sm/frnfluor.shtml
Ref 26. http://www.webmineral.com/specimens/picshow.php?id=3141&target=Fluorite#.W7KDboUXsws
Ref 27. https://www.pinterest.com/pin/111534528254934024/
Ref 29. https://www.pinterest.com/pin/592716000930774044/
Ref 30. https://www.crystalclassics.co.uk/events/1/43rd-gem-and-mineral-show-aue/
Ref 31. http://www.johnbetts-fineminerals.com/jhbnyc/mineralmuseum/picshow.php?id=24766
Ref 32. https://picclick.com/Fluorite-Asturias-Spain-372413243400.html
Ref 33. https://www.mineratminerals.com/archive/pink-fluorite-2c-la-mina-navidad-2c-mexico-detail



