Since ancient times jade[Ref1] has been used by artisans to create beautiful jewelry and works of art. Art objects of jade have been carved in China for more than 6000 years[Ref2] as exemplified by the jade dragon carved during the Zhou Dynasty (5th – 4th century BC) as shown in Figure 1. In Central America, the Mesoamerican cultures, the Olmecs, Mayans, and Aztecs[Ref3,4,5,6] prized jade for jewelry and religious objects as evidenced by the art objects in Figures 2-4. Since the arrival of the ancestors of the Maori in New Zealand in the 13th century[Ref7] jade, also called greenstone, or pounamu in the Maori language, has been imbued with spiritual significance and used to fabricate beautiful jewelry and objects such as shown in Figures 5-7[Ref8].
In this Blog I’ll describe those properties of the color and toughness of jade that underlie its long use as a gemstone in creating jewelry and artworks. I’ll also describe the current global sources of rough jade and jadeite and their geological origins. Also, in two subsequent Blogs I will describe first the long history jade as a gemstone, and present second a Gallery of jewelry and art works from around the ancient world.




Century[Ref13].

of a fishhook (Matau)[Ref 14,15].


April-May 1920[Ref14,17].

[Ref18].
JADE AS A GEMSTONE
Jade is a cultural term that encompasses a very durable silicate rock material which has been used in tool-making, sculpture, and jewelry, and other objects for over 5,000 years[Ref9]. Jade as a gemstone encompasses two rock types nephrite and jadetitite, the latter primarily comprised of the mineral jadeite[Ref1]. The amphiboles of the tremolite-actinolite series of minerals are the major components of nephrite jade. Mineral compositions of nephrite and Fen Sui Jadeite are described in Table I.
TABLE I. THE MINERAL COMPOSITIONS OF NEPHRITE AND FEN SUI JADEITE
| NEPHRITE [Ref19,20] | JADEITITE [Ref21,22,23,24,25] |
| Major Minerals | Major Minerals |
| Felted amphiboles of the tremolite-actinolite series | Jadeite-Kosmochlor mineral series, which are pyroxenes |
| Minerals in Minor Quantities | Mineral in Minor quantity |
| diopside, grossularitic garnet, magnetite, chromite, graphite, apatite, rutile, pyrite, datolite, vesuvianite, prehnite, talc, serpentine, and titanite. | Diopside, also a pyroxene |
Properties of Jade As a Gemstone
A number of the properties of jade underlie its wide use as a gemstone. Its many rich colors and its soft luster underlie its wide aesthetic use in jewelry and art objects. Its hardness and toughness allow its use in forms of jewelry susceptible to wear. In this section I’ll describe the sources of the various colors of nephrite and jadeite jades, and describe its physical properties underlying its use in its durable jewelry.
Sources of colors in nephrite jade
Absorption of visible light by iron, chromium, and manganese ions present in the members of the Tremolite-Amphibole mineral series which comprise nephrite jade underlies its colors. Results of a study to determine the identity of these metal ions that was made on over a broad sampling of colored nephrites from China have determined the color-metal-source relationships shown in Table 4 of Reference – as presented in Figure – below. The visible light absorption spectra of the colored specimens are shown in Figure 7 of the reference.

Among the mechanisms the electron (charge) transfer responsible for colors, elevation to a higher energy d-orbital of a transition metal ion and decay back to the ground level such as indicated by the excitation of an impurity manganese ion,
Mn3+ (4Eg)-> 5T2g and by the excitation and decay of a constituent iron ion, chromium ion Cr3+ (4A2) ->4T2,, or iron ion Fe3+ ( 6A1) -> 4E + 4A1(4G). Also electron (charge) transfer from an ion richer in electrons to one less rich such as between oxygen and iron ions O2- -> Fe3+, between iron ions Fe2+ -> Fe3+, and between iron and titanium ions Fe2+ -> Ti4+ can contribute absorption peaks to the absorption spectrum of nephrite jade. Annotated absorption spectra of nephrite jade specimens of various colors shown in Figure — of Reference– illustrate the relationship between absorption peaks and transmission windows within each spectrum[Ref25]..
Micron-sized iron oxide mineral impurities listed in TABLE – also can lend colors to nephrite jade, and also contribute to the yellow to black range of colors in jadeite.
TABLE II. IMPURITY MINERALS CONTRIBUTING TO YELL0W-BLACK COLOR RANGE IN NEPHRITE[Ref1]
| COLOR | MINERAL | FORMULA |
| Yellow | Limonite | FeO(OH) – nH2O, n is arbitrary |
| Red | Hematite | Fe2O3 |
| Black | Magnetite | Fe3O4 |
| Black | Chromite | FeCr2O4 |
| Brown | Combinations of Limonite, Hematite, and Magnetite | As above |
Sources of colors in jadeite
Iron, chromium, manganese, and silicon ions in the lattice of jadeite (NaAlSi2O6) are responsible for its green blue, and lavender colors as summarized in TABLE –[Ref1]. In jadeite with hues of green both the ferric Fe3+, chromicCr3+ ions substitute for aluminum ion Al3+. In blue and lavender jadeite ferrous, and ferric ion, Fe2+ and Fe3+, and the manganese III ions Mn3+ substitute for aluminum ions. The titanium ions Ti4+ substitute for the Silicon ion Si4.
Light absorption by the ferric ions in jadeite produces a peak in the blue at 437 nm and absorption by chromic ions results in peaks at in the 580-700 nm range[Ref1].
In blue and violet jadeite absorption of light by a ferrous ion which results in transfer an electrons through an oxygen site to a neighboring titanium ion (charge transfer) results in a wide absorption peak at 630 nm[Ref1,3].
Micron-sized iron oxide mineral impurities listed in TABLE – can lend colors to nephrite jade, and also can contribute to the yellow to black range of colors in jadeite.
TABLE III. TRANSITION METAL ION SOURCES OF COLOR IN NEPHRTE JADEITE
| COLOR | ELEMENT | ABSORPTION CENTER | REFERENCE |
| Green | Iron and Chromium | Cr3+, Fe3+ | Figure 9 in Ref 28. |
| Greyish Green | Iron | Fe3+ | Figure 13C in Ref 28. |
| Blue | Iron and Titanium | Fe3+, *Fe2+ -O- Ti4+ | Figure 12 in Ref 28. |
| Lavender (violet) | Iron, Manganese, and Titanium | Fe3+, Mn3+, *Fe2+-O-Ti4+ | Figure 13D in Ref 28. |
| Lavender (Purple) | Iron and Manganese | Fe3+, Mn3+ | Figure 13B in Ref 28. |
| Yellow, Brown, Red, Black | Iron, Oxygen, and Hydrogen | Fe2+, Fe3+, O2-, OH1- | 29 |

Strength properties of jade
The hardness of a material is its ability to resist abrasion; the toughness of a material is its ability absorb energy and plastically deform without fracturing under stress[Ref31]. These features underlie the use of nephrite in ancient times in fabricating durable tools such as the axe shown in Figure 10 of fabricated in China during the era of the Liangshu Culture in approximately 3300-2220 BC.The range of Mohs Hardness values of nephrite jade lie in the range of 6.0-6.5[Ref], and are less than those of jadeite, which lie in the range 6.5-7.0[Ref]. The hardness of both lie just below that of 7.0 for quartz[Ref].
In the 1973 study of Bradt, Newnham, and Biggers values of the fracture toughness and fracture strength of both nephrite and jadeite jade were determined with their measured values interpreted in terms of their microcrystalline structures[Ref38]. Comparison of the values of these quantities for both varieties of jade and with the values of fracture toughness for both quartz and corundum, both well-known as gemstones, are presented in TABLE .
TABLE IV. STRENGTH AND TOUGHNESS OF JADE AND OTHER GEMSTONES
| GEMSTONE | FRACTURE STRENGTH Dynes/cm-2 | FRACTURE SURFACE ENERGY Ergs/cm2 | FRACTURE TOUGHNESS Dynes/ cm-(3/2) |
| Nephrite | 2.22 x 109 | 2.26 x 105 | 7. 7 x 108 |
| Jadeite | 1.02 x 109 | 1.21 x 105 | 7.1 x 108 |
| Quartzite | —– | 4.32 x 103 | 7 x 107 |
| Alumina(Polycrystalline aluminum oxide, corundum) | —– | 1.5—5.0 x 105 | 3.5-4.4 x 108 |
| Quartz Crystal | —– | 1.03 x 103 | 5.0 x 107 |
| Corundum Crystal | —– | 6.0 x 10 2 | 7.0 x 107 |
Examination of the TABLE IV shows the values of fracture strength, fracture free energy, and fracture toughness of nephrite and jadeite to exceed those of alumina and quartzite, both also being polycrystalline materials, and far exceed those of single crystals of quartz and corundum, both also known to be durable gemstones.
The results show that propagation of cracks across crystals and along boundaries between crystals require considerably more energy than propagation along cleavage and parting planes in crystals. Propagation of cracks across and between crystals of jadeite and crack propagation along boundaries between elongated crystal grains and bundles of grains in nephrite are shown in Figures -,-,-. Both fibrous structure and trans-granular fracture impede crack propagation and toughen the material.
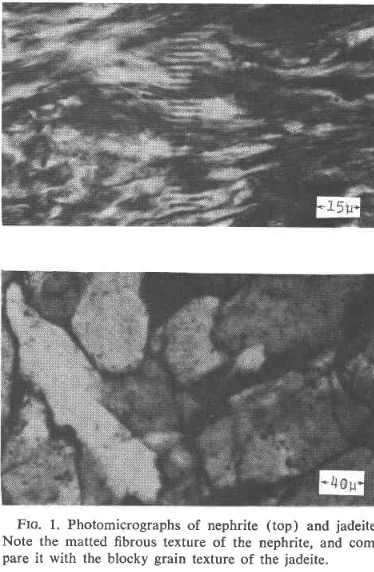
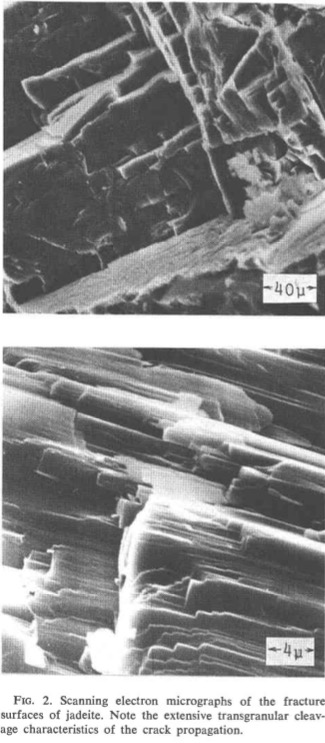
[Ref38].fracture.

amphibole crystals[Ref38].
Geology and Occurrences of Nephrite and Jadeitite
Both nephrite jade and jadeitite jade are found world wide as shown in the map of Figure 14.
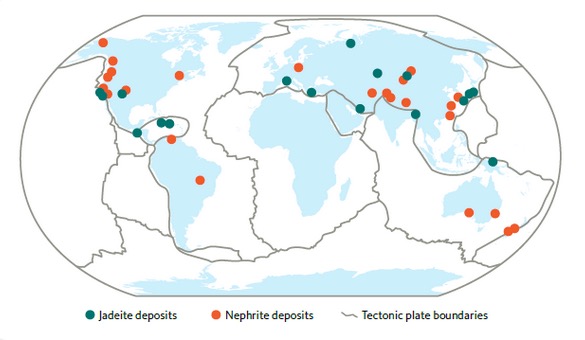
Among known world-wide sites of jade production, the main sources of nephrite production are in the United States and British Columbia[Ref39,40]; the main sources of Jadeite are in Myanmar, Russia, Central America, and Japan[Ref40].
Examples of jadeite rough and nephrite rough materials from current sources are shown in Figures 15 and 16.


The sources of both jadeite (jadeitite) and nephrite as shown in the map of Figure 14 are located along the boundaries of colliding tectonic plates comprising the earths lithosphere which underlie the continents and ocean beds and meet at boundaries at which the continental plates override the oceanic plates as shown in Figure 14[Ref39,43].
Geological processes in formation of jadeite
The geological processes of the formation of jadeite are summarized below in Figures 17-19.
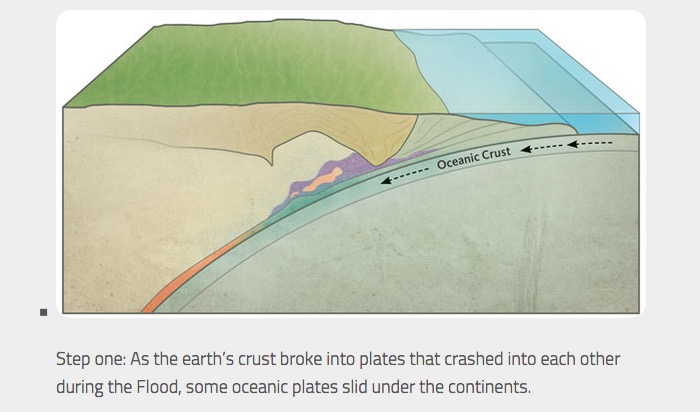

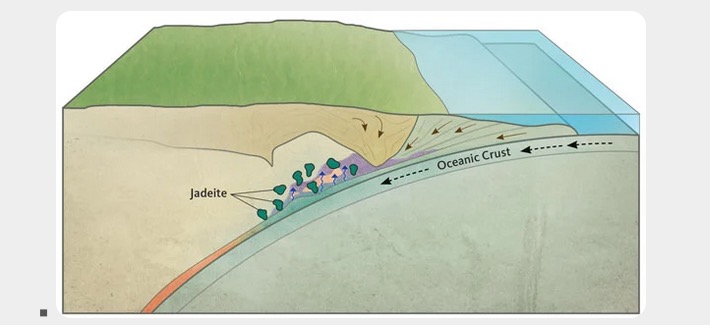
Geological processes in formation of nephrite jade
Most nephrite occurs along fault contacts between serpentinite [Ref46] and in basic to acidic igneous rocks or sandstone following obduction[Ref47] of the serpentinite body by the oceanic plate. It forms by the action of calcium- and silica-rich fluids on the serpentinite with the nephrite replacing the serpentinite. under low pressure-temperature conditions[Ref48].
Nephrite can also from in dolomitic marble from calcium- and silica-rich solutions from intruding molten magma of granite composition[Ref49].
REFERENCES JADE
Ref 1. https://www.mindat.org/min-10403.html
Ref 2. https://www.ancient.eu/article/1088/jade-in-ancient-china/
Ref 3. https://www.jadeite-atelier.com/blogs/jade-articles/history-of-jade-in-mesoamerica
Ref 4. https://www.ancient.eu/Olmec_Civilization/
Ref 5. https://www.ancient.eu/Maya_Civilization/
Ref 6. https://www.ancient.eu/Aztec_Civilization/
Ref 7. https://en.wikipedia.org/wiki/M%C4%81ori_people
Ref 8. https://teara.govt.nz/en/pounamu-jade-or-greenstone
Ref 9. https://www.ancient.eu/image/6778/zhou-dynasty-jade-dragon/
Ref 10. https://www.metmuseum.org/art/collection/search/310279
Ref 11 https://www.ancient.eu/Kinich_Janaab_Pacal/
Ref 12.
Ref 13. http://www.clevelandart.org/art/1966.361
Ref 14. https://www.mountainjade.co.nz/about-jade/greenstone-meanings-and-designs/
Ref 15. https://www.earthboundkiwi.com/shop-by-designs/nephrite-jade-maori-prosperity-fish-hook-necklace/
Ref 16. https://collections.tepapa.govt.nz/object/555878
Ref 17. https://collections.tepapa.govt.nz/object/555878
Ref 18. https://thestunzfamily.files.wordpress.com/2011/06/double-koru-pounamu1.jpg
Ref 19. https://www.mindat.org/min-10403.html
Ref 20. https://www.mindat.org/min-42720.html
Ref 21. https://www.mindat.org/min-40527.html
Ref 22. https://www.mindat.org/min-42720.html
Ref 23. https://www.mindat.org/min-2062.html
Ref 24. https://www.mindat.org/min-52021.html
Ref 26. http://geohavens.com/index.php?option=com_content&view=article&id=140&Itemid=649
Ref 27. https://www.jstage.jst.go.jp/article/jmps/111/5/111_151103/_pdf
Ref 28. https://www.gia.edu/gems-gemology/spring-2017-japanese-jadeite
Ref 29. http://geohavens.com/index.php?option=com_content&view=article&id=140&Itemid=649
Ref 30. https://en.wikipedia.org/wiki/Charge-transfer_complex
Ref 31. https://en.wikipedia.org/wiki/Toughness
Ref 33. https://en.wikipedia.org/wiki/Liangzhu_culture
Ref 34. https://en.wikipedia.org/wiki/Mohs_scale_of_mineral_hardness
Ref 35. https://en.wikipedia.org/wiki/Nephrite
Ref 36. https://en.wikipedia.org/wiki/Jadeite
Ref 37. https://en.wikipedia.org/wiki/Quartz
Ref 38. http://www.minsocam.org/ammin/AM58/AM58_727.pdf
Ref 39. https://ebcky.com/2018/09/16/jade-beauty-under-pressure/
Ref 40. https://www.eurojade.fr/en/british-columbia-nephrite
Ref 41. https://www.gia.edu/gia-news-research/witnessing-the-56th-myanma-gems-emporium
Ref 42. http://www.geologypage.com/2017/09/nephrite-jade-sources.html
Ref 43. https://en.wikipedia.org/wiki/Plate_tectonics
Ref 44. https://ebcky.com/2018/09/16/jade-beauty-under-pressure/
Ref 45. https://nsm.utdallas.edu/~rjstern/pdfs/SternGeology13.pdf
Ref 46. https://www.sandatlas.org/serpentinite/
Ref 47. https://link.springer.com/referenceworkentry/10.1007%2F3-540-31080-0_72
Ref 48.
Ref 49. https://www.sciencedirect.com/science/article/pii/S1674987118302329



