Today I want to talk about opal as a gemstone and it’s, not expected, biological role in grasses and algae.
Opal as a Gemstone
Gem-quality opals are characterized as Precious Opal, also called Fire Opals and Common Opals. Precious Opal as a gemstone is prized by collectors and can display all the colors of the rainbow, (Figures 1-4), or present only one (Figures 5 & 6). The differences in the presentations of colors lies in their structural and chemical differences.






As seen in [Ref 1], chemically, opal is a hydrated silicon dioxide with the formula SiO2-nH2O [Ref 2] in which “n” denotes a variable water content. In Fire Opal, (Opal AG), the weight percentage of water as estimated from Figure 3 in [Ref 2], is about 5%. Opal is not structured like Quartz, (SiO2), in which the silicon and two oxygen atoms are arranged in a fixed array throughout the crystal. In opal the silicon dioxide is amorphous with no crystalline order and forms spheres in the 10s-100s nanometers size-range.
In Fire Opal the spheres are typically arranged within a layer in a very nearly ordered fashion, either in a square or hexagonal array, as shown in Figure 7, in which both are in different regions within the layer. These layers, when stacked, present an orderly arrangement as shown in Figure 8.


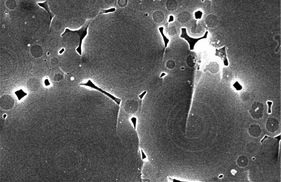
In Common Opals the spheres are not arranged in an orderly fashion, but are in a disorderly array as seen in Figure 9. And, in some instances, spheres with large differences in their diameters and with an orderly arrangement as seen in Figure 10. These features prevent light diffraction.

Source of Color in Preciouse Opal
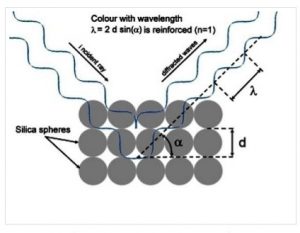
In Precious Opal the uniform diameters of the silica spheres result in a uniform thickness of the layers as seen in Figure 8. This structural feature of precious opal allows it to diffract light [Ref 6]. As in diagram below, the wavelength of the light given by the symbol λ which is diffracted depends directly on the distance the light travels between each layer as given in the diffraction equation: λ =2dsin(α) where d is the diameter of the spheres and α is both the angle that is incident and diffracted away [Ref 6]. The total distance traveled by both incident and diffracted light depends upon the angle α at which the total incident and diffracted light is travels and equals 2d sin(α). When the distances traveled by both their incident and diffracted light waves are equal they reinforce each other. This effect gives the “fire” in the color as no other color is selected. When the light strikes perpendicular to the layers λ = 2d. When the light strikes at any other angle α, the distance traveled is lessened by the factor sin(α) and the corresponding wavelength which is diffracted is larger.
In Precious Opals the uniformity of the diameter of the spheres and in various regions of the gem the spacing of the spheres is not necessarily uniform over the entire volume of the opal. These factors will result in the diffraction of different wavelengths over the surface of the gem as evident in Figures 1-4.
Sources of color in Fire Opal
In Fire Opal, a variety of Common Opal, the diameters of the spheres are in the range o5 to 25 nanometers. Spheres this size being only about one-tenth that of visible light do not diffract and do not present the play of colors as does Precious Opal; they possess the disordered structural features of Common Opal as shown in Figures 9 & 10. Instead the color is uniform over the volume of the opal, and is imparted by inclusion of the iron compound Fe2O3 as a pigment [Ref 7]. As examples: yellow, orange, red, & brown Common Opals of Mexico have nanometer-sized iron inclusions. Similarly the blue-colored opals from Peru do not diffract and are colored by a copper silicate mineral [Ref 7].
Opals in Biology
Having made a transition from the material sciences to biophysics along my career path, I became intrigued with the interrelationships between the worlds of the inorganic and the organic. Among these interrelationships are the roles of minerals in biological organisms. In addition to the vital roles of the elements such as iron, iodine, calcium, potassium, sodium, phosphorous, in the biology of the cell and in organism, minerals also play important roles as skeletal structural elements across the animal and plant kingdoms. The roles of these structural elements are to support and/or protect the soft tissues and organs of the organism.
How Opal Protects the Cell Walls of Grasses and Diatoms
As an example of opal in biology I will describe the mechanisms whereby opal serves to protect grasses and the soft cellular components of diatoms, a major group of microalgae.
In grasses
Grasses, by virtue of their environments, are subject to attack and consumption by both insects and grazing animals. As a protective mechanism, leaf, stem, and roots of many grasses have adapted by hardening their outermost layer of tissue, the epidermis [Ref 8 & 9]. Hardening of the tissue is achieved through deposition of hydrated silica (SiO2-nH2O) in the walls of the epidermal cells [Ref 8]. The scanning electron micrograph shown in Figure 11, [Ref 10] show the deposition of silica in the walls of grass cells and forming rows of stony phytoliths [Ref 10]. The phytoliths, arranged in rows, provide hardened regions along the length of leaf, stem and root. The phytoliths deter attack by herbivores by reducing the digestibility of the grass tissue, and in insects, they exact wear on their mandibles. The jagged shapes of phytoliths in grasses, such as those in sorghum, [Ref 10], also suggest that the grasses eaten by grazing animals may be an irritant to the soft tissues of the mouth.

In diatoms
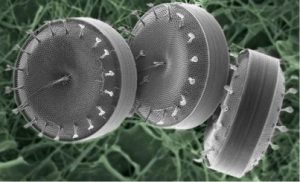
Diatoms are single-cell microalgae and are among the most common of plant plankton, distributed ocean-wide and in fresh waters. Their species exhibit a myriad of shapes, some quite beautiful. (Figure 12) [Ref 11]

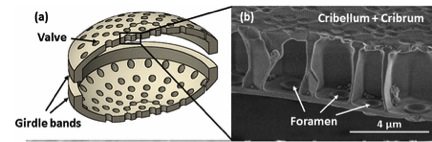
Their cell walls, of hydrated silica, both protect the soft contents of the cell and allow the in-flux of nutrients. They also provide a route for the out-flux of gases and waste products [Ref 11]. The cell wall comprises two cap-like halves known as frustures as shown in the examples in Figure 13. The outer appearance of the cell wall is one of a ribbed structure with numerous pores distributed between the ribs.
The pores overlie a chamber, as seen in Figure 13-b. The innermost surface of the cell wall lies against the outer membrane of the cell where they are taken inside the cell via the membrane of the cell. The small pores allow entrapment of nutrients within the chamber and act as valves to protect against loss of nutrients on the occasion of lessened concentrations of nutrients outside the cell wall. [Ref 13]. This provides the ability to compete with nearby feeding diatoms.
Ref 1. https://www.gia.edu/opal-description
Ref 2. https://www.mindat.org/min-3004.html
Ref 3. https://www.tandfonline.com/doi/abs/10.1080/00167617108728743
Ref 4. https://www.blackstaropal.com/pages/opalessence-about-opal-the-queen-of-gems
Ref 5. http://nhminsci.blogspot.com/2012/07/loving-ethiopian-opals.html
Ref 6. http://www.uniqueopals.ch/opal-play-of-colour.htm
Ref 7. http://www.ingentaconnect.com/contentone/asp/jctn/2016/00000013/00000003/art00080
Ref 8. https://www.ncbi.nlm.nih.gov/pubmed/16638012
Ref 9. http://wfu.me/andersonlab/data/role-of-silica-in-serengeti-grasses-2/
Ref 10. http://rivkaelbaum.wixsite.com/rivka-elbaum/silica
Ref 11. https://en.wikipedia.org/wiki/Diatom
Ref 12. http://science.jrank.org/pages/2051/Diatoms.html
Ref 13. http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0059548



